
By Nicholas Banadda
The first initial consignment of 19,200 doses of Lanacapavir from GlobalfUND has arrived in Uganda, the ministry of health statement issued stated.
According to the statement issued by ministry of health, the drug is administered every six months to prevent HIV among persons with substantial risk of acquiring HIV.
According to the ministry statement the drugs will be distributed to high burden and high incidence districts effective March 2026.
In statement issued last month by the Uganda Aids Commission (UAC) , announced that a long-acting HIV prevention drug, Lenacapavir, was proven to provide 100% protection to HIV-negative individuals when used as pre-exposure prophylaxis (PrEP)
The breakthrough follows successful global clinical trials in which Ugandan scientists played a key role.
In a press release statement issued, the Commission stated: “Research on the use of Lenacapavir for pre-exposure prophylaxis (PrEP) has successfully established that this six-monthly injection offers 100% protection to HIV-negative individuals against acquiring HIV.”
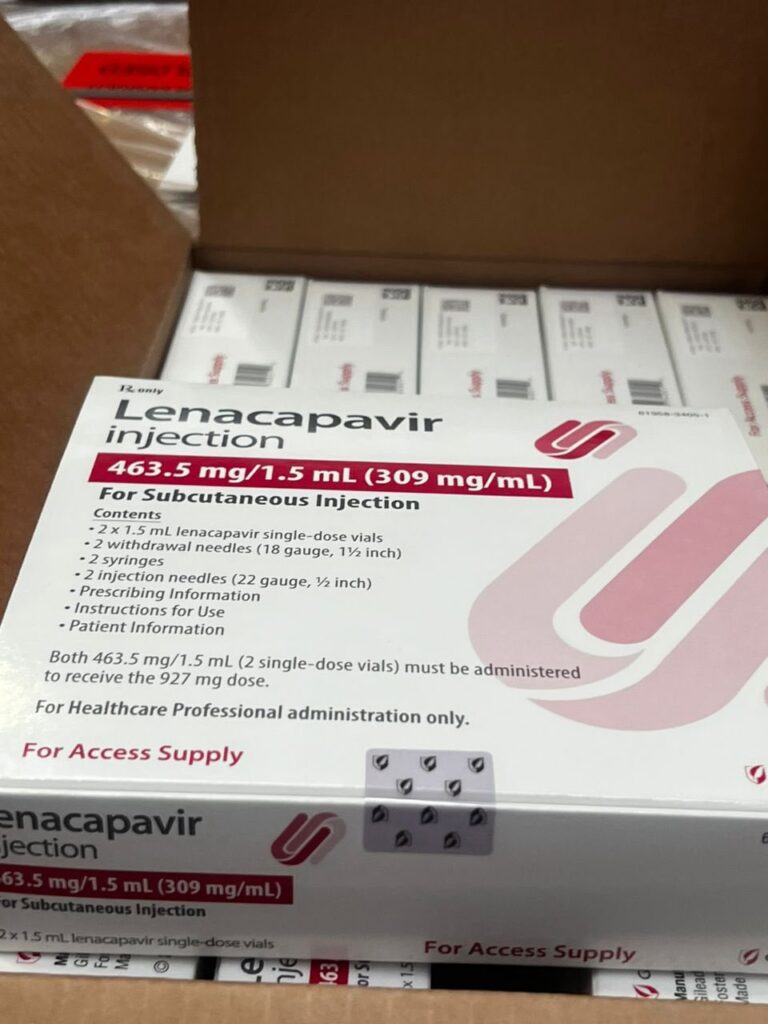
The development comes after the drug’s production and use were approved by the US Food and Drug Administration (FDA). Uganda now plans to incorporate Lenacapavir into its national HIV prevention strategy.
The Ministry of Health lead the coordination, in collaboration with stakeholders, to evaluate and licensed the drug through the National Drug Authority.
“The process will also include incorporating Lenacapavir into the national essential medicines list and revising data collection tools to track its use effectively. Once these steps have been completed, Lenacapavir will be integrated into Uganda’s HIV prevention program, expanding options for individuals at high risk of HIV infection,” the statement states.
Until then, Uganda will continue offering existing PrEP options. ends